Androgen receptor
| Androgen_recep | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 crystal structure of the human androgen receptor ligand binding domain bound with an androgen receptor nh2-terminal peptide, ar20-30, and r1881 | |||||||||
| Identifiers | |||||||||
| Symbol | Androgen_recep | ||||||||
| Pfam | PF02166 | ||||||||
| InterPro | IPR001103 | ||||||||
| |||||||||
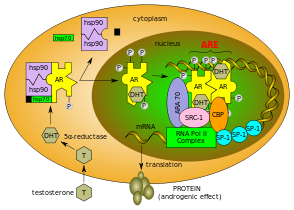
The androgen receptor (AR), also known as NR3C4 (nuclear receptor subfamily 3, group C, member 4), is a type of nuclear receptor[9] that is activated by binding any of the androgenic hormones, including testosterone and dihydrotestosterone,[10] in the cytoplasm and then translocating into the nucleus. The androgen receptor is most closely related to the progesterone receptor, and progestins in higher dosages can block the androgen receptor.[11][12]
The main function of the androgen receptor is as a DNA-binding transcription factor that regulates gene expression;[13] however, the androgen receptor has other functions as well.[14] Androgen-regulated genes are critical for the development and maintenance of the male sexual phenotype.
Function
[edit]Effect on development
[edit]In some cell types, testosterone interacts directly with androgen receptors, whereas, in others, testosterone is converted by 5-alpha-reductase to dihydrotestosterone, an even more potent agonist for androgen receptor activation.[15] Testosterone appears to be the primary androgen receptor-activating hormone in the Wolffian duct, whereas dihydrotestosterone is the main androgenic hormone in the urogenital sinus, urogenital tubercle, and hair follicles.[16] Testosterone is therefore responsible primarily for the development of male primary sexual characteristics, whilst dihydrotestosterone is responsible for secondary male characteristics.
Androgens cause slow maturation of the bones, but more of the potent maturation effect comes from the estrogen produced by aromatization of androgens. Steroid users of teen age may find that their growth had been stunted by androgen and/or estrogen excess. People with too little sex hormones can be short during puberty but end up taller as adults as in androgen insensitivity syndrome or estrogen insensitivity syndrome.[17]
Knockout-mice studies have shown that the androgen receptor is essential for normal female fertility, being required for development and full functionality of the ovarian follicles and ovulation, working through both intra-ovarian and neuroendocrine mechanisms.[18]
Maintenance of male skeletal integrity
[edit]Via the androgen receptor, androgens play a key role in the maintenance of male skeletal integrity. The regulation of this integrity by androgen receptor (AR) signaling can be attributed to both osteoblasts and osteocytes.[19]
Role in females
[edit]The AR plays a role in regulating female sexual, somatic, and behavioral functions. Experimental data using AR knockout female mice, provides evidence that the promotion of cardiac growth, kidney hypertrophy, cortical bone growth and regulation of trabecular bone structure is a result of DNA-binding-dependent actions of the AR in females.
Moreover, the importance of understanding female androgen receptors lies in their role in several genetic disorders including androgen insensitivity syndrome (AIS). Complete (CAIS) and partial (PAIS) which are a result of mutations in the genes that code for AR. These mutations cause the inactivation of AR due to mutations conferring resistance to circulating testosterone, with more than 400 different AR mutations reported.[citation needed]
Mechanism of action
[edit]Genomic
[edit]The primary mechanism of action for androgen receptors is direct regulation of gene transcription.
Androgens (also called androgenic hormones), such as testosterone or dihydrotestosterone, are understood to exert their primary effects through binding to an androgen receptor in the cytosol. The receptor is translocated to the nucleus upon androgen binding and ultimately results in the transcriptional regulation of a number of genes via androgen responsive elements.[20] This androgen response mechanism is perhaps best known and characterized in the context of male sexual differentiation and puberty, but plays a role in a variety of tissue types and processes.[21][22] Upon binding to androgens, the androgen receptor dissociates from accessory proteins, translocates into the nucleus, dimerizes, and then stimulates transcription of androgen-responsive genes.[23]
The binding of an androgen to the androgen receptor results in a conformational change in the receptor that, in turn, causes dissociation of heat shock proteins, transport from the cytosol into the cell nucleus, and dimerization. The androgen receptor dimer binds to a specific sequence of DNA known as a hormone response element, where it forms macromolacular protein condensates that might facilitate rapid gene regulation as consequence of local high protein concentrations together with other coregulators.[24] Androgen receptors interact with other proteins in the nucleus, resulting in up- or down-regulation of specific gene transcription.[25] Up-regulation or activation of transcription results in increased synthesis of messenger RNA, which, in turn, is translated by ribosomes to produce specific proteins. One of the known target genes of androgen receptor activation is the insulin-like growth factor 1 receptor (IGF-1R).[26] Thus, changes in levels of specific proteins in cells is one way that androgen receptors control cell behavior.
One function of androgen receptor that is independent of direct binding to its target DNA sequence is facilitated by recruitment via other DNA-binding proteins. One example is serum response factor, a protein that activates several genes that cause muscle growth.[27]
Androgen receptor is modified by post-translational modification through acetylation,[28] which directly promotes AR-mediated transactivation, apoptosis[29] and contact-independent growth of prostate cancer cells.[30] AR acetylation is induced by androgens[31] and determines recruitment into chromatin.[32] The AR acetylation site is a key target of NAD-dependent and TSA-dependent histone deacetylases[33] and long non-coding RNA.[34]
Non-genomic
[edit]More recently, androgen receptors have been shown to have a second mode of action. As has been also found for other steroid hormone receptors such as estrogen receptors, androgen receptors can have actions that are independent of their interactions with DNA.[14][35] Androgen receptors interact with certain signal transduction proteins in the cytoplasm. Androgen binding to cytoplasmic androgen receptors can cause rapid changes in cell function independent of changes in gene transcription, such as changes in ion transport. Regulation of signal transduction pathways by cytoplasmic androgen receptors can indirectly lead to changes in gene transcription, for example, by leading to phosphorylation of other transcription factors.
Genetics
[edit]Gene
[edit]In humans, the androgen receptor is encoded by the AR gene located on the X chromosome at Xq11–12.[36][37]
Deficiencies
[edit]At least 165 disease-causing mutations in this gene have been discovered.[38] The androgen insensitivity syndrome, formerly known as testicular feminization, is caused by a mutation in the androgen receptor gene on the X chromosome (locus: Xq11–Xq12).[39] The androgen receptor seems to affect neuron physiology and is defective in Kennedy's disease.[40][41] In addition, point mutations and trinucleotide repeat polymorphisms have been linked to a number of additional disorders.[42]
CAG repeats
[edit]The AR gene contains CAG repeats that affect receptor function, where fewer repeats leads to increased receptor sensitivity to circulating androgens and more repeats leads to decreased receptor sensitivity. Studies have shown that racial variation in CAG repeats exists,[43][44] with African-Americans having fewer repeats than non-Hispanic white Americans.[43] The racial trends in CAG repeats parallels the incidence and mortality of prostate cancer in these two groups.
Mutations
[edit]The enhancer and the gene encoding for these receptors contain recurrent mutations, such as structural rearrangements and copy number changes, acquired in the progression of metastatic castration-resistant prostate cancer (mCRPC) treatment with therapy targeting these receptors (abiraterone, enzalutamide), make the disease progression determined by the androgen receptor genotype.[45]
Structure
[edit]

Isoforms
[edit]Two isoforms of the androgen receptor (A and B) have been identified:[47]
- AR-A – 87 kDa; N-terminus truncated (lacks the first 187 amino acids), which results from in vitro proteolysis.[48]
- AR-B – 110 kDa; full length
Domains
[edit]Like other nuclear receptors, the androgen receptor is modular in structure and is composed of the following functional domains labeled A through F:[49]
- A/B) – N-terminal is a regulatory domain which is intrinsically disordered[50] and contains several key motifs/segments:[51]
- Dimerization surface involving residues 1–36, the 23FQNLF27 motif[52] (B only) nd 370–494, both of which interact with the ligand binding domain (LBD) in an intramolecular[53][54][55] head-to-tail interaction[56][57][58]
- FXXLF motif; where F = phenylalanine, L = leucine, and X = any amino acid residue a
- Polyglutamine repeat[59] (AR-B only)
- Transactivation unit 1 (TAU-1) between residues 101 and 370 required for full ligand-activated transcriptional activity [60]
- Transactivation unit 5 (TAU-5) between residues 360–485 is responsible for the constitutive activity (activity without bound ligand)[60]
- Dimerization surface involving residues 1–36, the 23FQNLF27 motif[52] (B only) nd 370–494, both of which interact with the ligand binding domain (LBD) in an intramolecular[53][54][55] head-to-tail interaction[56][57][58]
Despite being highly disordered, the NTD domain adopts a 3D conformation in which two dynamically compact regions form, an N-terminal region (NR) and a C-terminal region (CR), each having distinct topological dynamics and function.[46]
- C) – DNA binding domain (DBD)
- Contains two Zinc fingers[61]
- D) – Hinge region; flexible region that connects the DBD with the LBD; along with the DBD, contains a ligand dependent nuclear localization signal[62]
- E) – Ligand binding domain (LBD) containing
- activation function 2 (AF-2), responsible for agonist induced activity (activity in the presence of bound agonist)
- AF-2 binds either the N-terminal FXXFL motif intramolecularly or coactivator proteins (containing the LXXLL or preferably FXXFL motifs)[58]
- A ligand dependent nuclear export signal[63]
- F) – C-terminal domain
Splice variants
[edit]AR-V7 is an androgen receptor splice variant that can be detected in circulating tumor cells of metastatic prostate cancer patients[64][65] and is predictive of resistance to some drugs (caused by missing Ligand-binding domain in this splice variant).[66]
Clinical significance
[edit]High expression in androgen receptor has been linked to aggression and sex drive by affecting the HPA and HPG axis[67]
Aberrant androgen receptor coregulator activity may contribute to the progression of prostate cancer.[68][45]
Ligands
[edit]| Compound | RBA[b] |
|---|---|
| Metribolone | 100 |
| Dihydrotestosterone | 85 |
| Cyproterone acetate | 7.8 |
| Bicalutamide | 1.4 |
| Nilutamide | 0.9 |
| Hydroxyflutamide | 0.57 |
| Flutamide | <0.0057 |
Notes:
| |
Agonists
[edit]- Endogenous androgens (e.g., testosterone, dihydrotestosterone, androstenedione, androstenediol, dehydroepiandrosterone)
- Synthetic androgens (e.g., methyltestosterone, metandienone, nandrolone, trenbolone, oxandrolone, stanozolol)
Mixed
[edit]Antagonists
[edit]- Steroidal antiandrogens (e.g., cyproterone acetate, chlormadinone acetate, spironolactone, oxendolone)
- Nonsteroidal antiandrogens (e.g., flutamide, nilutamide, bicalutamide, enzalutamide, apalutamide, RU-58841)
- N-Terminal domain antiandrogens (e.g., bisphenol A, EPI-001, ralaniten, JN compounds)[70]
As a drug target
[edit]The AR is an important therapeutic target in prostate cancer. Thus many different antiandrogens have been developed, primarily targeting the ligand-binding domain of the protein.[71] AR ligands can either be classified based on their structure (steroidal or nonsteroidal) or based on their ability to activate or inhibit transcription (agonists or antagonists).[72] Inhibitors that target alternative functional domains (N-terminal domain, DNA-binding domain) of the protein are still under development.[70]
Drug resistance
[edit]Alteration of ARs may lead to treatment resistance (castration resistance) in prostate cancer as there may be missense mutations of the ligand binding domain, amplifications of the gene coding for this receptor or in its enhancer, mostly, suggesting the presence of different subclones with different genotypes of these receptors.[45]
Interactions
[edit]Androgen receptor has been shown to interact with:
- AKT1,[73]
- BAG1,[74][75][76]
- Beta-catenin,[77][78][79][80][81][82]
- BRCA1,[83][84]
- C-jun,[85]
- Calmodulin 1,[86]
- Caveolin 1,[87]
- CDK9,[88]
- COX5B,[89]
- CREB-binding protein,[90][91][92][93]
- Cyclin D1,[94][95][96][97]
- Cyclin-dependent kinase 7,[98]
- DACH1,[99]
- Death associated protein 6,[100]
- L-DOPA,[101]
- EFCAB6,[102]
- Epidermal growth factor receptor,[103][104]
- FOXO1,[105]
- GAPDH,[106]
- Gelsolin,[107]
- GNB2L1,[108]
- GSK3B,[109]
- HDAC1,[110]
- HSP90AA1,[111][112]
- HTATIP,[110]
- MAGEA11,[113][114]
- MED1,[115]
- MYST2,[116]
- NCOA1,[78][117][118]
- NCOA2,[77][92][113][119][120]
- NCOA3,[119][121][122]
- NCOA4,[73][120][123][124][125][126][127][128][129]
- NCOA6,[130]
- NCOR2,[77][131][132]
- NONO,[92]
- p300,[133]
- PA2G4,[134]
- PAK6,[135][136]
- PATZ1,[137]
- PIAS2,[138][139]
- PRPF6,[140]
- PTEN,[141]
- RAD9A,[142]
- RANBP9,[143]
- RCHY1,[144]
- Retinoblastoma protein,[145][146]
- RNF14,[120][123][147][148]
- RNF4,[137][149][150]
- SART3,[151]
- SIRT1,[33]
- SMAD3,[152][153][154]
- Small heterodimer partner,[155]
- Src,[141][156][157]
- SRY,[158]
- STAT3,[159][160]
- SVIL,[161]
- Testicular receptor 2,[162]
- Testicular receptor 4,[163]
- TGFB1I1,[123][164]
- TMF1,[165]
- TRIM68,[166]
- UBE2I,[77][78][167][168][169][170]
- UXT,[171] and
- ZMIZ1.[172]
See also
[edit]References
[edit]- ^ a b c GRCh38: Ensembl release 89: ENSG00000169083 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000046532 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Quigley CA, De Bellis A, Marschke KB, el-Awady MK, Wilson EM, French FS (June 1995). "Androgen receptor defects: historical, clinical, and molecular perspectives". Endocrine Reviews. 16 (3): 271–321. doi:10.1210/edrv-16-3-271. PMID 7671849.
- ^ Gottlieb B, Lombroso R, Beitel LK, Trifiro MA (January 2005). "Molecular pathology of the androgen receptor in male (in)fertility". Reproductive Biomedicine Online. 10 (1): 42–8. doi:10.1016/S1472-6483(10)60802-4. PMID 15705293.
- ^ Choong CS, Wilson EM (December 1998). "Trinucleotide repeats in the human androgen receptor: a molecular basis for disease". Journal of Molecular Endocrinology. 21 (3): 235–57. doi:10.1677/jme.0.0210235. PMID 9845666.
- ^ Meehan KL, Sadar MD (May 2003). "Androgens and androgen receptor in prostate and ovarian malignancies". Frontiers in Bioscience. 8 (1–3): d780–800. doi:10.2741/1063. PMID 12700055.
- ^ Lu NZ, Wardell SE, Burnstein KL, Defranco D, Fuller PJ, Giguere V, et al. (December 2006). "International Union of Pharmacology. LXV. The pharmacology and classification of the nuclear receptor superfamily: glucocorticoid, mineralocorticoid, progesterone, and androgen receptors". Pharmacological Reviews. 58 (4): 782–97. doi:10.1124/pr.58.4.9. PMID 17132855. S2CID 28626145.
- ^ Roy AK, Lavrovsky Y, Song CS, Chen S, Jung MH, Velu NK, et al. (1999). Regulation of androgen action. Vitamins & Hormones. Vol. 55. Elsevier. pp. 309–52. doi:10.1016/S0083-6729(08)60938-3. ISBN 978-0-12-709855-5. PMID 9949684.
- ^ Bardin CW, Brown T, Isomaa VV, Jänne OA (1983). "Progestins can mimic, inhibit and potentiate the actions of androgens". Pharmacology & Therapeutics. 23 (3): 443–59. doi:10.1016/0163-7258(83)90023-2. PMID 6371845.
- ^ Raudrant D, Rabe T (2003). "Progestogens with antiandrogenic properties". Drugs. 63 (5): 463–92. doi:10.2165/00003495-200363050-00003. PMID 12600226. S2CID 28436828.
- ^ Mooradian AD, Morley JE, Korenman SG (February 1987). "Biological actions of androgens". Endocrine Reviews. 8 (1): 1–28. doi:10.1210/edrv-8-1-1. PMID 3549275.
- ^ a b Heinlein CA, Chang C (October 2002). "The roles of androgen receptors and androgen-binding proteins in nongenomic androgen actions". Molecular Endocrinology. 16 (10): 2181–7. doi:10.1210/me.2002-0070. PMID 12351684.
- ^ Davison SL, Bell R (April 2006). "Androgen physiology". Seminars in Reproductive Medicine. 24 (2): 71–7. doi:10.1055/s-2006-939565. PMID 16633980. S2CID 260321076.
- ^ Sinisi AA, Pasquali D, Notaro A, Bellastella A (2003). "Sexual differentiation". Journal of Endocrinological Investigation. 26 (3 Suppl): 23–28. PMID 12834017.
- ^ Frank GR (September 2003). "Role of estrogen and androgen in pubertal skeletal physiology". Medical and Pediatric Oncology. 41 (3): 217–21. doi:10.1002/mpo.10340. PMID 12868122.
- ^ Walters KA, Simanainen U, Handelsman DJ (March 2010). "Molecular insights into androgen actions in male and female reproductive function from androgen receptor knockout models". Human Reproduction Update. 16 (5): 543–58. doi:10.1093/humupd/dmq003. PMID 20231167.
- ^ Sinnesael M, Claessens F, Laurent M, Dubois V, Boonen S, Deboel L, et al. (December 2012). "Androgen receptor (AR) in osteocytes is important for the maintenance of male skeletal integrity: evidence from targeted AR disruption in mouse osteocytes". Journal of Bone and Mineral Research. 27 (12): 2535–43. doi:10.1002/jbmr.1713. PMID 22836391.
- ^ Gelmann EP (July 2002). "Molecular biology of the androgen receptor". Journal of Clinical Oncology. 20 (13): 3001–3015. doi:10.1200/JCO.2002.10.018. PMID 12089231.
- ^ Li J, Al-Azzawi F (June 2009). "Mechanism of androgen receptor action". Maturitas. 63 (2): 142–148. doi:10.1016/j.maturitas.2009.03.008. PMID 19372015.
- ^ Masiutin M, Yadav M (2023). "Alternative androgen pathways". WikiJournal of Medicine. 10: X. doi:10.15347/WJM/2023.003. S2CID 257943362.
- ^ Coffey K, Robson CN (November 2012). "Regulation of the androgen receptor by post-translational modifications". The Journal of Endocrinology. 215 (2): 221–237. doi:10.1530/JOE-12-0238. PMID 22872761. S2CID 20933176.
- ^ Yavuz S, Kabbech H (October 2023). "Compartmentalization of androgen receptors at endogenous genes in living cells". Nucleic Acids Research. 51 (20): 10992–11009. doi:10.1093/nar/gkad803. PMC 10639085. PMID 37791849.
- ^ Heemers HV, Tindall DJ (December 2007). "Androgen receptor (AR) coregulators: a diversity of functions converging on and regulating the AR transcriptional complex". Endocrine Reviews. 28 (7): 778–808. doi:10.1210/er.2007-0019. PMID 17940184.
- ^ Pandini G, Mineo R, Frasca F, Roberts CT, Marcelli M, Vigneri R, et al. (March 2005). "Androgens up-regulate the insulin-like growth factor-I receptor in prostate cancer cells". Cancer Research. 65 (5): 1849–57. doi:10.1158/0008-5472.CAN-04-1837. PMID 15753383.
- ^ Vlahopoulos S, Zimmer WE, Jenster G, Belaguli NS, Balk SP, Brinkmann AO, et al. (March 2005). "Recruitment of the androgen receptor via serum response factor facilitates expression of a myogenic gene". The Journal of Biological Chemistry. 280 (9): 7786–92. doi:10.1074/jbc.M413992200. PMID 15623502.
- ^ Fu M, Wang C, Reutens AT, Wang J, Angeletti RH, Siconolfi-Baez L, et al. (July 2000). "p300 and p300/cAMP-response element-binding protein-associated factor acetylate the androgen receptor at sites governing hormone-dependent transactivation". The Journal of Biological Chemistry. 275 (27): 20853–60. doi:10.1074/jbc.M000660200. PMID 10779504.
- ^ Fu M, Wang C, Wang J, Zhang X, Sakamaki T, Yeung YG, et al. (May 2002). "Androgen receptor acetylation governs trans activation and MEKK1-induced apoptosis without affecting in vitro sumoylation and trans-repression function". Molecular and Cellular Biology. 22 (10): 3373–88. doi:10.1128/mcb.22.10.3373-3388.2002. PMC 133781. PMID 11971970.
- ^ Fu M, Rao M, Wang C, Sakamaki T, Wang J, Di Vizio D, et al. (December 2003). "Acetylation of androgen receptor enhances coactivator binding and promotes prostate cancer cell growth". Molecular and Cellular Biology. 23 (23): 8563–75. doi:10.1128/mcb.23.23.8563-8575.2003. PMC 262657. PMID 14612401.
- ^ Gong J, Zhu J, Goodman OB, Pestell RG, Schlegel PN, Nanus DM, et al. (March 2006). "Activation of p300 histone acetyltransferase activity and acetylation of the androgen receptor by bombesin in prostate cancer cells". Oncogene. 25 (14): 2011–21. doi:10.1038/sj.onc.1209231. PMID 16434977.
- ^ Fu M, Rao M, Wu K, Wang C, Zhang X, Hessien M, et al. (July 2004). "The androgen receptor acetylation site regulates cAMP and AKT but not ERK-induced activity". The Journal of Biological Chemistry. 279 (28): 29436–49. doi:10.1074/jbc.M313466200. PMID 15123687.
- ^ a b Fu M, Liu M, Sauve AA, Jiao X, Zhang X, Wu X, et al. (November 2006). "Hormonal control of androgen receptor function through SIRT1". Molecular and Cellular Biology. 26 (21): 8122–35. doi:10.1128/MCB.00289-06. PMC 1636736. PMID 16923962.
- ^ Yang L, Lin C, Jin C, Yang JC, Tanasa B, Li W, et al. (August 2013). "lncRNA-dependent mechanisms of androgen-receptor-regulated gene activation programs". Nature. 500 (7464): 598–602. Bibcode:2013Natur.500..598Y. doi:10.1038/nature12451. PMC 4034386. PMID 23945587.
- ^ Fix C, Jordan C, Cano P, Walker WH (July 2004). "Testosterone activates mitogen-activated protein kinase and the cAMP response element binding protein transcription factor in Sertoli cells". Proceedings of the National Academy of Sciences of the United States of America. 101 (30): 10919–24. Bibcode:2004PNAS..10110919F. doi:10.1073/pnas.0404278101. PMC 503720. PMID 15263086.
- ^ Chang CS, Kokontis J, Liao ST (April 1988). "Molecular cloning of human and rat complementary DNA encoding androgen receptors". Science. 240 (4850): 324–6. Bibcode:1988Sci...240..324C. doi:10.1126/science.3353726. PMID 3353726.
- ^ Trapman J, Klaassen P, Kuiper GG, van der Korput JA, Faber PW, van Rooij HC, et al. (May 1988). "Cloning, structure and expression of a cDNA encoding the human androgen receptor". Biochemical and Biophysical Research Communications. 153 (1): 241–8. doi:10.1016/S0006-291X(88)81214-2. PMID 3377788.
- ^ Šimčíková D, Heneberg P (December 2019). "Refinement of evolutionary medicine predictions based on clinical evidence for the manifestations of Mendelian diseases". Scientific Reports. 9 (1): 18577. Bibcode:2019NatSR...918577S. doi:10.1038/s41598-019-54976-4. PMC 6901466. PMID 31819097.
- ^ Brown TR (1995). "Human androgen insensitivity syndrome". Journal of Andrology. 16 (4): 299–303. doi:10.1002/j.1939-4640.1995.tb00533.x. PMID 8537246. S2CID 25940392.
- ^ Kennedy WR, Alter M, Sung JH (July 1968). "Progressive proximal spinal and bulbar muscular atrophy of late onset. A sex-linked recessive trait". Neurology. 18 (7): 671–80. doi:10.1212/WNL.18.7.671. PMID 4233749. S2CID 45735233.
- ^ Yu Z, Dadgar N, Albertelli M, Gruis K, Jordan C, Robins DM, et al. (October 2006). "Androgen-dependent pathology demonstrates myopathic contribution to the Kennedy disease phenotype in a mouse knock-in model". The Journal of Clinical Investigation. 116 (10): 2663–72. doi:10.1172/JCI28773. PMC 1564432. PMID 16981011.
- ^ Rajender S, Singh L, Thangaraj K (March 2007). "Phenotypic heterogeneity of mutations in androgen receptor gene". Asian Journal of Andrology. 9 (2): 147–79. doi:10.1111/j.1745-7262.2007.00250.x. PMID 17334586.
- ^ a b Sartor O, Zheng Q, Eastham JA (February 1999). "Androgen receptor gene CAG repeat length varies in a race-specific fashion in men without prostate cancer". Urology. 53 (2): 378–80. doi:10.1016/s0090-4295(98)00481-6. PMID 9933058.
- ^ Weintrob N, Eyal O, Slakman M, Segev Becker A, Israeli G, Kalter-Leibovici O, et al. (2018). "The effect of CAG repeats length on differences in hirsutism among healthy Israeli women of different ethnicities". PLOS ONE. 13 (3): e0195046. Bibcode:2018PLoSO..1395046W. doi:10.1371/journal.pone.0195046. PMC 5871002. PMID 29584789.
- ^ a b c Herberts C, Annala M, Sipola J, Ng SW, Chen XE, Nurminen A, et al. (August 2022). "Deep whole-genome ctDNA chronology of treatment-resistant prostate cancer". Nature. 608 (7921): 199–208. Bibcode:2022Natur.608..199H. doi:10.1038/s41586-022-04975-9. PMID 35859180. S2CID 250730778.
- ^ a b Sheikhhassani V, Scalvini B, Ng J, Heling LW, Ayache Y, Evers TM, et al. (June 2022). "Topological dynamics of an intrinsically disordered N-terminal domain of the human androgen receptor". Protein Science. 31 (6): e4334. doi:10.1002/pro.4334. PMID 35634773.
- ^ Wilson CM, McPhaul MJ (February 1994). "A and B forms of the androgen receptor are present in human genital skin fibroblasts". Proceedings of the National Academy of Sciences of the United States of America. 91 (4): 1234–8. Bibcode:1994PNAS...91.1234W. doi:10.1073/pnas.91.4.1234. PMC 43131. PMID 8108393.
- ^ Gregory CW, He B, Wilson EM (December 2001). "The putative androgen receptor-A form results from in vitro proteolysis". Journal of Molecular Endocrinology. 27 (3): 309–19. doi:10.1677/jme.0.0270309. PMID 11719283.
- ^ Brinkmann AO, Klaasen P, Kuiper GG, van der Korput JA, Bolt J, de Boer W, et al. (1989). "Structure and function of the androgen receptor". Urological Research. 17 (2): 87–93. doi:10.1007/BF00262026. PMID 2734982. S2CID 19706366.
- ^ Zhang F, Biswas M, Massah S, Lee J, Lingadahalli S, Wong S, et al. (January 2023). "Dynamic phase separation of the androgen receptor and its coactivators key to regulate gene expression". Nucleic Acids Research. 51 (1): 99–116. doi:10.1093/nar/gkac1158. PMC 9841400. PMID 36535377.
- ^ Jenster G, van der Korput HA, Trapman J, Brinkmann AO (March 1995). "Identification of two transcription activation units in the N-terminal domain of the human androgen receptor". The Journal of Biological Chemistry. 270 (13): 7341–7346. doi:10.1074/jbc.270.13.7341. hdl:1765/8551. PMID 7706276.
- ^ Asangani I, Blair IA, Van Duyne G, Hilser VJ, Moiseenkova-Bell V, Plymate S, et al. (2021). "Using biochemistry and biophysics to extinguish androgen receptor signaling in prostate cancer". The Journal of Biological Chemistry. 296: 100240. doi:10.1074/jbc.REV120.012411. PMC 7949100. PMID 33384381.
- ^ Schaufele F, Carbonell X, Guerbadot M, Borngraeber S, Chapman MS, Ma AA, et al. (July 2005). "The structural basis of androgen receptor activation: intramolecular and intermolecular amino-carboxy interactions". Proceedings of the National Academy of Sciences of the United States of America. 102 (28): 9802–9807. Bibcode:2005PNAS..102.9802S. doi:10.1073/pnas.0408819102. PMC 1168953. PMID 15994236.
- ^ Klokk TI, Kurys P, Elbi C, Nagaich AK, Hendarwanto A, Slagsvold T, et al. (March 2007). "Ligand-specific dynamics of the androgen receptor at its response element in living cells". Molecular and Cellular Biology. 27 (5): 1823–1843. doi:10.1128/MCB.01297-06. PMC 1820481. PMID 17189428.
- ^ van Royen ME, Cunha SM, Brink MC, Mattern KA, Nigg AL, Dubbink HJ, et al. (April 2007). "Compartmentalization of androgen receptor protein-protein interactions in living cells". The Journal of Cell Biology. 177 (1): 63–72. doi:10.1083/jcb.200609178. PMC 2064112. PMID 17420290.
- ^ Langley E, Zhou ZX, Wilson EM (December 1995). "Evidence for an anti-parallel orientation of the ligand-activated human androgen receptor dimer". The Journal of Biological Chemistry. 270 (50): 29983–29990. doi:10.1074/jbc.270.50.29983. PMID 8530400.
- ^ Berrevoets CA, Doesburg P, Steketee K, Trapman J, Brinkmann AO (August 1998). "Functional interactions of the AF-2 activation domain core region of the human androgen receptor with the amino-terminal domain and with the transcriptional coactivator TIF2 (transcriptional intermediary factor2)". Molecular Endocrinology. 12 (8): 1172–1183. doi:10.1210/mend.12.8.0153. hdl:1765/8892. PMID 9717843.
- ^ a b Dubbink HJ, Hersmus R, Verma CS, van der Korput HA, Berrevoets CA, van Tol J, et al. (September 2004). "Distinct recognition modes of FXXLF and LXXLL motifs by the androgen receptor". Molecular Endocrinology. 18 (9): 2132–2150. doi:10.1210/me.2003-0375. PMID 15178743.
- ^ Van-Duyne G, Blair IA, Sprenger C, Moiseenkova-Bell V, Plymate S, Penning TM (2023-01-01). Litwack G (ed.). "The androgen receptor". Vitamins and Hormones. Hormone Receptors: Structures and Functions. 123. Academic Press: 439–481. doi:10.1016/bs.vh.2023.01.001. ISBN 978-0-443-13455-5. PMID 37717994.
- ^ a b Callewaert L, Van Tilborgh N, Claessens F (January 2006). "Interplay between two hormone-independent activation domains in the androgen receptor". Cancer Research. 66 (1): 543–553. doi:10.1158/0008-5472.CAN-05-2389. PMID 16397271.
- ^ Tan MH, Li J, Xu HE, Melcher K, Yong EL (January 2015). "Androgen receptor: structure, role in prostate cancer and drug discovery". Acta Pharmacologica Sinica. 36 (1): 3–23. doi:10.1038/aps.2014.18. PMC 4571323. PMID 24909511.
- ^ Kaku N, Matsuda K, Tsujimura A, Kawata M (August 2008). "Characterization of nuclear import of the domain-specific androgen receptor in association with the importin alpha/beta and Ran-guanosine 5'-triphosphate systems". Endocrinology. 149 (8): 3960–9. doi:10.1210/en.2008-0137. PMC 2488236. PMID 18420738.
- ^ Saporita AJ, Zhang Q, Navai N, Dincer Z, Hahn J, Cai X, et al. (October 2003). "Identification and characterization of a ligand-regulated nuclear export signal in androgen receptor". The Journal of Biological Chemistry. 278 (43): 41998–2005. doi:10.1074/jbc.M302460200. PMID 12923188.
- ^ Silberstein JL, Taylor MN, Antonarakis ES (April 2016). "Novel Insights into Molecular Indicators of Response and Resistance to Modern Androgen-Axis Therapies in Prostate Cancer". Current Urology Reports. 17 (4): 29. doi:10.1007/s11934-016-0584-4. PMC 4888068. PMID 26902623.
- ^ Antonarakis ES, Lu C, Wang H, Luber B, Nakazawa M, Roeser JC, et al. (September 2014). "AR-V7 and resistance to enzalutamide and abiraterone in prostate cancer". The New England Journal of Medicine. 371 (11): 1028–38. doi:10.1056/NEJMoa1315815. PMC 4201502. PMID 25184630.
- ^ "Biomarker-Driven Therapy With Nivolumab and Ipilimumab in Treating Patients With Metastatic Hormone-Resistant Prostate Cancer Expressing AR-V7 - Full Text View - ClinicalTrials.gov". clinicaltrials.gov. Retrieved 2016-02-27.
- ^ Cunningham RL, Lumia AR, McGinnis MY (2012). "Androgen receptors, sex behavior, and aggression". Neuroendocrinology. 96 (2): 131–40. doi:10.1159/000337663. PMC 3474193. PMID 22414851.
- ^ Heinlein CA, Chang C (April 2002). "Androgen receptor (AR) coregulators: an overview". Endocrine Reviews. 23 (2): 175–200. doi:10.1210/edrv.23.2.0460. PMID 11943742.
- ^ Ayub M, Levell MJ (August 1989). "The effect of ketoconazole related imidazole drugs and antiandrogens on [3H] R 1881 binding to the prostatic androgen receptor and [3H]5 alpha-dihydrotestosterone and [3H]cortisol binding to plasma proteins". J. Steroid Biochem. 33 (2): 251–5. doi:10.1016/0022-4731(89)90301-4. PMID 2788775.
- ^ a b Elshan ND, Rettig MB, Jung ME (22 November 2018). "Molecules targeting the androgen receptor (AR) signaling axis beyond the AR-Ligand binding domain". Medicinal Research Reviews. 39 (3): 910–960. doi:10.1002/med.21548. PMC 6608750. PMID 30565725.
- ^ Helsen C, Van den Broeck T, Voet A, Prekovic S, Van Poppel H, Joniau S, et al. (August 2014). "Androgen receptor antagonists for prostate cancer therapy". Endocrine-Related Cancer. 21 (4): T105–18. doi:10.1530/ERC-13-0545. PMID 24639562.
- ^ Gao W, Bohl CE, Dalton JT (September 2005). "Chemistry and structural biology of androgen receptor". Chemical Reviews. 105 (9): 3352–70. doi:10.1021/cr020456u. PMC 2096617. PMID 16159155.
- ^ a b Lin HK, Yeh S, Kang HY, Chang C (June 2001). "Akt suppresses androgen-induced apoptosis by phosphorylating and inhibiting androgen receptor". Proceedings of the National Academy of Sciences of the United States of America. 98 (13): 7200–5. Bibcode:2001PNAS...98.7200L. doi:10.1073/pnas.121173298. PMC 34646. PMID 11404460.
- ^ Shatkina L, Mink S, Rogatsch H, Klocker H, Langer G, Nestl A, et al. (October 2003). "The cochaperone Bag-1L enhances androgen receptor action via interaction with the NH2-terminal region of the receptor". Molecular and Cellular Biology. 23 (20): 7189–97. doi:10.1128/MCB.23.20.7189-7197.2003. PMC 230325. PMID 14517289.
- ^ Knee DA, Froesch BA, Nuber U, Takayama S, Reed JC (April 2001). "Structure-function analysis of Bag1 proteins. Effects on androgen receptor transcriptional activity". The Journal of Biological Chemistry. 276 (16): 12718–24. doi:10.1074/jbc.M010841200. PMID 11278763.
- ^ Froesch BA, Takayama S, Reed JC (May 1998). "BAG-1L protein enhances androgen receptor function". The Journal of Biological Chemistry. 273 (19): 11660–6. doi:10.1074/jbc.273.19.11660. PMID 9565586.
- ^ a b c d Song LN, Coghlan M, Gelmann EP (January 2004). "Antiandrogen effects of mifepristone on coactivator and corepressor interactions with the androgen receptor". Molecular Endocrinology. 18 (1): 70–85. doi:10.1210/me.2003-0189. PMID 14593076.
- ^ a b c Masiello D, Chen SY, Xu Y, Verhoeven MC, Choi E, Hollenberg AN, et al. (October 2004). "Recruitment of beta-catenin by wild-type or mutant androgen receptors correlates with ligand-stimulated growth of prostate cancer cells". Molecular Endocrinology. 18 (10): 2388–401. doi:10.1210/me.2003-0436. PMID 15256534.
- ^ Yang F, Li X, Sharma M, Sasaki CY, Longo DL, Lim B, et al. (March 2002). "Linking beta-catenin to androgen-signaling pathway". The Journal of Biological Chemistry. 277 (13): 11336–44. doi:10.1074/jbc.M111962200. PMID 11792709.
- ^ Amir AL, Barua M, McKnight NC, Cheng S, Yuan X, Balk SP (August 2003). "A direct beta-catenin-independent interaction between androgen receptor and T cell factor 4". The Journal of Biological Chemistry. 278 (33): 30828–34. doi:10.1074/jbc.M301208200. PMID 12799378.
- ^ Mulholland DJ, Read JT, Rennie PS, Cox ME, Nelson CC (August 2003). "Functional localization and competition between the androgen receptor and T-cell factor for nuclear beta-catenin: a means for inhibition of the Tcf signaling axis". Oncogene. 22 (36): 5602–13. doi:10.1038/sj.onc.1206802. PMID 12944908.
- ^ Pawlowski JE, Ertel JR, Allen MP, Xu M, Butler C, Wilson EM, et al. (June 2002). "Liganded androgen receptor interaction with beta-catenin: nuclear co-localization and modulation of transcriptional activity in neuronal cells". The Journal of Biological Chemistry. 277 (23): 20702–10. doi:10.1074/jbc.M200545200. PMID 11916967.
- ^ Park JJ, Irvine RA, Buchanan G, Koh SS, Park JM, Tilley WD, et al. (November 2000). "Breast cancer susceptibility gene 1 (BRCAI) is a coactivator of the androgen receptor". Cancer Research. 60 (21): 5946–9. PMID 11085509.
- ^ Yeh S, Hu YC, Rahman M, Lin HK, Hsu CL, Ting HJ, et al. (October 2000). "Increase of androgen-induced cell death and androgen receptor transactivation by BRCA1 in prostate cancer cells". Proceedings of the National Academy of Sciences of the United States of America. 97 (21): 11256–61. Bibcode:2000PNAS...9711256Y. doi:10.1073/pnas.190353897. PMC 17187. PMID 11016951.
- ^ Sato N, Sadar MD, Bruchovsky N, Saatcioglu F, Rennie PS, Sato S, et al. (July 1997). "Androgenic induction of prostate-specific antigen gene is repressed by protein-protein interaction between the androgen receptor and AP-1/c-Jun in the human prostate cancer cell line LNCaP". The Journal of Biological Chemistry. 272 (28): 17485–94. doi:10.1074/jbc.272.28.17485. PMID 9211894.
- ^ Cifuentes E, Mataraza JM, Yoshida BA, Menon M, Sacks DB, Barrack ER, et al. (January 2004). "Physical and functional interaction of androgen receptor with calmodulin in prostate cancer cells". Proceedings of the National Academy of Sciences of the United States of America. 101 (2): 464–9. Bibcode:2004PNAS..101..464C. doi:10.1073/pnas.0307161101. PMC 327170. PMID 14695896.
- ^ Lu ML, Schneider MC, Zheng Y, Zhang X, Richie JP (April 2001). "Caveolin-1 interacts with androgen receptor. A positive modulator of androgen receptor mediated transactivation". The Journal of Biological Chemistry. 276 (16): 13442–51. doi:10.1074/jbc.M006598200. PMID 11278309.
- ^ Lee DK, Duan HO, Chang C (March 2001). "Androgen receptor interacts with the positive elongation factor P-TEFb and enhances the efficiency of transcriptional elongation". The Journal of Biological Chemistry. 276 (13): 9978–84. doi:10.1074/jbc.M002285200. PMID 11266437.
- ^ Beauchemin AM, Gottlieb B, Beitel LK, Elhaji YA, Pinsky L, Trifiro MA (2001). "Cytochrome c oxidase subunit Vb interacts with human androgen receptor: a potential mechanism for neurotoxicity in spinobulbar muscular atrophy". Brain Research Bulletin. 56 (3–4): 285–97. doi:10.1016/S0361-9230(01)00583-4. PMID 11719263. S2CID 24740136.
- ^ Kim J, Jia L, Stallcup MR, Coetzee GA (February 2005). "The role of protein kinase A pathway and cAMP responsive element-binding protein in androgen receptor-mediated transcription at the prostate-specific antigen locus". Journal of Molecular Endocrinology. 34 (1): 107–18. doi:10.1677/jme.1.01701. PMID 15691881.
- ^ Frønsdal K, Engedal N, Slagsvold T, Saatcioglu F (November 1998). "CREB binding protein is a coactivator for the androgen receptor and mediates cross-talk with AP-1". The Journal of Biological Chemistry. 273 (48): 31853–9. doi:10.1074/jbc.273.48.31853. PMID 9822653.
- ^ a b c Ishitani K, Yoshida T, Kitagawa H, Ohta H, Nozawa S, Kato S (July 2003). "p54nrb acts as a transcriptional coactivator for activation function 1 of the human androgen receptor". Biochemical and Biophysical Research Communications. 306 (3): 660–5. doi:10.1016/S0006-291X(03)01021-0. PMID 12810069.
- ^ Aarnisalo P, Palvimo JJ, Jänne OA (March 1998). "CREB-binding protein in androgen receptor-mediated signaling". Proceedings of the National Academy of Sciences of the United States of America. 95 (5): 2122–7. Bibcode:1998PNAS...95.2122A. doi:10.1073/pnas.95.5.2122. PMC 19270. PMID 9482849.
- ^ Reutens AT, Watanabe G, Albanese C, McPhaul MJ, Balk SP, Pestell RG (1998). "Cyclin D1 binds activating mutants of the androgen receptor". US Endocrine Society Meeting (1–528).
- ^ Reutens AT, Fu M, Wang C, Albanese C, McPhaul MJ, Sun Z, et al. (May 2001). "Cyclin D1 binds the androgen receptor and regulates hormone-dependent signaling in a p300/CBP-associated factor (P/CAF)-dependent manner". Molecular Endocrinology. 15 (5): 797–811. doi:10.1210/mend.15.5.0641. PMID 11328859.
- ^ Petre-Draviam CE, Williams EB, Burd CJ, Gladden A, Moghadam H, Meller J, et al. (January 2005). "A central domain of cyclin D1 mediates nuclear receptor corepressor activity". Oncogene. 24 (3): 431–44. doi:10.1038/sj.onc.1208200. PMID 15558026.
- ^ Knudsen KE, Cavenee WK, Arden KC (May 1999). "D-type cyclins complex with the androgen receptor and inhibit its transcriptional transactivation ability". Cancer Research. 59 (10): 2297–301. PMID 10344732.
- ^ Lee DK, Duan HO, Chang C (March 2000). "From androgen receptor to the general transcription factor TFIIH. Identification of cdk activating kinase (CAK) as an androgen receptor NH(2)-terminal associated coactivator". The Journal of Biological Chemistry. 275 (13): 9308–13. doi:10.1074/jbc.275.13.9308. PMID 10734072.
- ^ Wu K, Katiyar S, Witkiewicz A, Li A, McCue P, Song LN, et al. (April 2009). "The cell fate determination factor dachshund inhibits androgen receptor signaling and prostate cancer cellular growth". Cancer Research. 69 (8): 3347–55. doi:10.1158/0008-5472.CAN-08-3821. PMC 2669850. PMID 19351840.
- ^ Lin DY, Fang HI, Ma AH, Huang YS, Pu YS, Jenster G, et al. (December 2004). "Negative modulation of androgen receptor transcriptional activity by Daxx". Molecular and Cellular Biology. 24 (24): 10529–41. doi:10.1128/MCB.24.24.10529-10541.2004. PMC 533990. PMID 15572661.
- ^ Wafa LA, Cheng H, Rao MA, Nelson CC, Cox M, Hirst M, et al. (October 2003). "Isolation and identification of L-dopa decarboxylase as a protein that binds to and enhances transcriptional activity of the androgen receptor using the repressed transactivator yeast two-hybrid system". The Biochemical Journal. 375 (Pt 2): 373–83. doi:10.1042/BJ20030689. PMC 1223690. PMID 12864730.
- ^ Niki T, Takahashi-Niki K, Taira T, Iguchi-Ariga SM, Ariga H (February 2003). "DJBP: a novel DJ-1-binding protein, negatively regulates the androgen receptor by recruiting histone deacetylase complex, and DJ-1 antagonizes this inhibition by abrogation of this complex". Molecular Cancer Research. 1 (4): 247–61. PMID 12612053.
- ^ Bonaccorsi L, Carloni V, Muratori M, Formigli L, Zecchi S, Forti G, et al. (October 2004). "EGF receptor (EGFR) signaling promoting invasion is disrupted in androgen-sensitive prostate cancer cells by an interaction between EGFR and androgen receptor (AR)" (PDF). International Journal of Cancer. 112 (1): 78–86. doi:10.1002/ijc.20362. hdl:2158/395766. PMID 15305378. S2CID 46121331.
- ^ Bonaccorsi L, Muratori M, Carloni V, Marchiani S, Formigli L, Forti G, et al. (August 2004). "The androgen receptor associates with the epidermal growth factor receptor in androgen-sensitive prostate cancer cells". Steroids. 69 (8–9): 549–52. doi:10.1016/j.steroids.2004.05.011. hdl:2158/395763. PMID 15288768. S2CID 23831527.
- ^ Li P, Lee H, Guo S, Unterman TG, Jenster G, Bai W (January 2003). "AKT-independent protection of prostate cancer cells from apoptosis mediated through complex formation between the androgen receptor and FKHR". Molecular and Cellular Biology. 23 (1): 104–18. doi:10.1128/MCB.23.1.104-118.2003. PMC 140652. PMID 12482965.
- ^ Koshy B, Matilla T, Burright EN, Merry DE, Fischbeck KH, Orr HT, et al. (September 1996). "Spinocerebellar ataxia type-1 and spinobulbar muscular atrophy gene products interact with glyceraldehyde-3-phosphate dehydrogenase". Human Molecular Genetics. 5 (9): 1311–8. doi:10.1093/hmg/5.9.1311. PMID 8872471.
- ^ Nishimura K, Ting HJ, Harada Y, Tokizane T, Nonomura N, Kang HY, et al. (August 2003). "Modulation of androgen receptor transactivation by gelsolin: a newly identified androgen receptor coregulator". Cancer Research. 63 (16): 4888–94. PMID 12941811.
- ^ Rigas AC, Ozanne DM, Neal DE, Robson CN (November 2003). "The scaffolding protein RACK1 interacts with androgen receptor and promotes cross-talk through a protein kinase C signaling pathway". The Journal of Biological Chemistry. 278 (46): 46087–93. doi:10.1074/jbc.M306219200. PMID 12958311.
- ^ Wang L, Lin HK, Hu YC, Xie S, Yang L, Chang C (July 2004). "Suppression of androgen receptor-mediated transactivation and cell growth by the glycogen synthase kinase 3 beta in prostate cells". The Journal of Biological Chemistry. 279 (31): 32444–52. doi:10.1074/jbc.M313963200. PMID 15178691.
- ^ a b Gaughan L, Logan IR, Cook S, Neal DE, Robson CN (July 2002). "Tip60 and histone deacetylase 1 regulate androgen receptor activity through changes to the acetylation status of the receptor". The Journal of Biological Chemistry. 277 (29): 25904–13. doi:10.1074/jbc.M203423200. PMID 11994312.
- ^ Veldscholte J, Berrevoets CA, Brinkmann AO, Grootegoed JA, Mulder E (March 1992). "Anti-androgens and the mutated androgen receptor of LNCaP cells: differential effects on binding affinity, heat-shock protein interaction, and transcription activation". Biochemistry. 31 (8): 2393–9. doi:10.1021/bi00123a026. PMID 1540595.
- ^ Nemoto T, Ohara-Nemoto Y, Ota M (September 1992). "Association of the 90-kDa heat shock protein does not affect the ligand-binding ability of androgen receptor". The Journal of Steroid Biochemistry and Molecular Biology. 42 (8): 803–12. doi:10.1016/0960-0760(92)90088-Z. PMID 1525041. S2CID 24978960.
- ^ a b Bai S, He B, Wilson EM (February 2005). "Melanoma antigen gene protein MAGE-11 regulates androgen receptor function by modulating the interdomain interaction". Molecular and Cellular Biology. 25 (4): 1238–57. doi:10.1128/MCB.25.4.1238-1257.2005. PMC 548016. PMID 15684378.
- ^ Bai S, Wilson EM (March 2008). "Epidermal-growth-factor-dependent phosphorylation and ubiquitinylation of MAGE-11 regulates its interaction with the androgen receptor". Molecular and Cellular Biology. 28 (6): 1947–63. doi:10.1128/MCB.01672-07. PMC 2268407. PMID 18212060.
- ^ Wang Q, Sharma D, Ren Y, Fondell JD (November 2002). "A coregulatory role for the TRAP-mediator complex in androgen receptor-mediated gene expression". The Journal of Biological Chemistry. 277 (45): 42852–8. doi:10.1074/jbc.M206061200. PMID 12218053.
- ^ Sharma M, Zarnegar M, Li X, Lim B, Sun Z (November 2000). "Androgen receptor interacts with a novel MYST protein, HBO1". The Journal of Biological Chemistry. 275 (45): 35200–8. doi:10.1074/jbc.M004838200. PMID 10930412.
- ^ Ueda T, Mawji NR, Bruchovsky N, Sadar MD (October 2002). "Ligand-independent activation of the androgen receptor by interleukin-6 and the role of steroid receptor coactivator-1 in prostate cancer cells". The Journal of Biological Chemistry. 277 (41): 38087–94. doi:10.1074/jbc.M203313200. PMID 12163482.
- ^ Bevan CL, Hoare S, Claessens F, Heery DM, Parker MG (December 1999). "The AF1 and AF2 domains of the androgen receptor interact with distinct regions of SRC1". Molecular and Cellular Biology. 19 (12): 8383–92. doi:10.1128/mcb.19.12.8383. PMC 84931. PMID 10567563.
- ^ a b Wang Q, Udayakumar TS, Vasaitis TS, Brodie AM, Fondell JD (April 2004). "Mechanistic relationship between androgen receptor polyglutamine tract truncation and androgen-dependent transcriptional hyperactivity in prostate cancer cells". The Journal of Biological Chemistry. 279 (17): 17319–28. doi:10.1074/jbc.M400970200. PMID 14966121.
- ^ a b c He B, Wilson EM (March 2003). "Electrostatic modulation in steroid receptor recruitment of LXXLL and FXXLF motifs". Molecular and Cellular Biology. 23 (6): 2135–50. doi:10.1128/MCB.23.6.2135-2150.2003. PMC 149467. PMID 12612084.
- ^ Tan JA, Hall SH, Petrusz P, French FS (September 2000). "Thyroid receptor activator molecule, TRAM-1, is an androgen receptor coactivator". Endocrinology. 141 (9): 3440–50. doi:10.1210/endo.141.9.7680. PMID 10965917.
- ^ Gnanapragasam VJ, Leung HY, Pulimood AS, Neal DE, Robson CN (December 2001). "Expression of RAC 3, a steroid hormone receptor co-activator in prostate cancer". British Journal of Cancer. 85 (12): 1928–36. doi:10.1054/bjoc.2001.2179. PMC 2364015. PMID 11747336.
- ^ a b c He B, Minges JT, Lee LW, Wilson EM (March 2002). "The FXXLF motif mediates androgen receptor-specific interactions with coregulators". The Journal of Biological Chemistry. 277 (12): 10226–35. doi:10.1074/jbc.M111975200. PMID 11779876.
- ^ Alen P, Claessens F, Schoenmakers E, Swinnen JV, Verhoeven G, Rombauts W, et al. (January 1999). "Interaction of the putative androgen receptor-specific coactivator ARA70/ELE1alpha with multiple steroid receptors and identification of an internally deleted ELE1beta isoform". Molecular Endocrinology. 13 (1): 117–28. doi:10.1210/mend.13.1.0214. PMID 9892017.
- ^ Yeh S, Chang C (May 1996). "Cloning and characterization of a specific coactivator, ARA70, for the androgen receptor in human prostate cells". Proceedings of the National Academy of Sciences of the United States of America. 93 (11): 5517–21. Bibcode:1996PNAS...93.5517Y. doi:10.1073/pnas.93.11.5517. PMC 39278. PMID 8643607.
- ^ Miyamoto H, Yeh S, Wilding G, Chang C (June 1998). "Promotion of agonist activity of antiandrogens by the androgen receptor coactivator, ARA70, in human prostate cancer DU145 cells". Proceedings of the National Academy of Sciences of the United States of America. 95 (13): 7379–84. Bibcode:1998PNAS...95.7379M. doi:10.1073/pnas.95.13.7379. PMC 22623. PMID 9636157.
- ^ Yeh S, Lin HK, Kang HY, Thin TH, Lin MF, Chang C (May 1999). "From HER2/Neu signal cascade to androgen receptor and its coactivators: a novel pathway by induction of androgen target genes through MAP kinase in prostate cancer cells". Proceedings of the National Academy of Sciences of the United States of America. 96 (10): 5458–63. Bibcode:1999PNAS...96.5458Y. doi:10.1073/pnas.96.10.5458. PMC 21881. PMID 10318905.
- ^ Zhou ZX, He B, Hall SH, Wilson EM, French FS (February 2002). "Domain interactions between coregulator ARA(70) and the androgen receptor (AR)". Molecular Endocrinology. 16 (2): 287–300. doi:10.1210/mend.16.2.0765. PMID 11818501.
- ^ Gao T, Brantley K, Bolu E, McPhaul MJ (October 1999). "RFG (ARA70, ELE1) interacts with the human androgen receptor in a ligand-dependent fashion, but functions only weakly as a coactivator in cotransfection assays". Molecular Endocrinology. 13 (10): 1645–56. doi:10.1210/mend.13.10.0352. PMID 10517667.
- ^ Goo YH, Na SY, Zhang H, Xu J, Hong S, Cheong J, et al. (February 2004). "Interactions between activating signal cointegrator-2 and the tumor suppressor retinoblastoma in androgen receptor transactivation". The Journal of Biological Chemistry. 279 (8): 7131–5. doi:10.1074/jbc.M312563200. PMID 14645241.
- ^ Liao G, Chen LY, Zhang A, Godavarthy A, Xia F, Ghosh JC, et al. (February 2003). "Regulation of androgen receptor activity by the nuclear receptor corepressor SMRT". The Journal of Biological Chemistry. 278 (7): 5052–61. doi:10.1074/jbc.M206374200. PMID 12441355.
- ^ Dotzlaw H, Moehren U, Mink S, Cato AC, Iñiguez Lluhí JA, Baniahmad A (April 2002). "The amino terminus of the human AR is target for corepressor action and antihormone agonism". Molecular Endocrinology. 16 (4): 661–73. doi:10.1210/mend.16.4.0798. PMID 11923464.
- ^ Fu M, Wang C, Reutens AT, Wang J, Angeletti RH, Siconolfi-Baez L, et al. (July 2000). "p300 and p300/cAMP-response element-binding protein-associated factor acetylate the androgen receptor at sites governing hormone-dependent transactivation". The Journal of Biological Chemistry. 275 (27): 20853–60. doi:10.1074/jbc.M000660200. PMID 10779504.
- ^ Zhang Y, Fondell JD, Wang Q, Xia X, Cheng A, Lu ML, et al. (August 2002). "Repression of androgen receptor mediated transcription by the ErbB-3 binding protein, Ebp1". Oncogene. 21 (36): 5609–18. doi:10.1038/sj.onc.1205638. PMID 12165860.
- ^ Yang F, Li X, Sharma M, Zarnegar M, Lim B, Sun Z (May 2001). "Androgen receptor specifically interacts with a novel p21-activated kinase, PAK6". The Journal of Biological Chemistry. 276 (18): 15345–53. doi:10.1074/jbc.M010311200. PMID 11278661.
- ^ Lee SR, Ramos SM, Ko A, Masiello D, Swanson KD, Lu ML, et al. (January 2002). "AR and ER interaction with a p21-activated kinase (PAK6)". Molecular Endocrinology. 16 (1): 85–99. doi:10.1210/mend.16.1.0753. PMID 11773441.
- ^ a b Pero R, Lembo F, Palmieri EA, Vitiello C, Fedele M, Fusco A, et al. (February 2002). "PATZ attenuates the RNF4-mediated enhancement of androgen receptor-dependent transcription". The Journal of Biological Chemistry. 277 (5): 3280–5. doi:10.1074/jbc.M109491200. PMID 11719514.
- ^ Kotaja N, Aittomäki S, Silvennoinen O, Palvimo JJ, Jänne OA (December 2000). "ARIP3 (androgen receptor-interacting protein 3) and other PIAS (protein inhibitor of activated STAT) proteins differ in their ability to modulate steroid receptor-dependent transcriptional activation". Molecular Endocrinology. 14 (12): 1986–2000. doi:10.1210/mend.14.12.0569. PMID 11117529.
- ^ Moilanen AM, Karvonen U, Poukka H, Yan W, Toppari J, Jänne OA, et al. (February 1999). "A testis-specific androgen receptor coregulator that belongs to a novel family of nuclear proteins". The Journal of Biological Chemistry. 274 (6): 3700–4. doi:10.1074/jbc.274.6.3700. PMID 9920921.
- ^ Zhao Y, Goto K, Saitoh M, Yanase T, Nomura M, Okabe T, et al. (August 2002). "Activation function-1 domain of androgen receptor contributes to the interaction between subnuclear splicing factor compartment and nuclear receptor compartment. Identification of the p102 U5 small nuclear ribonucleoprotein particle-binding protein as a coactivator for the receptor". The Journal of Biological Chemistry. 277 (33): 30031–9. doi:10.1074/jbc.M203811200. PMID 12039962.
- ^ a b Lin HK, Hu YC, Lee DK, Chang C (October 2004). "Regulation of androgen receptor signaling by PTEN (phosphatase and tensin homolog deleted on chromosome 10) tumor suppressor through distinct mechanisms in prostate cancer cells". Molecular Endocrinology. 18 (10): 2409–23. doi:10.1210/me.2004-0117. PMID 15205473.
- ^ Wang L, Hsu CL, Ni J, Wang PH, Yeh S, Keng P, et al. (March 2004). "Human checkpoint protein hRad9 functions as a negative coregulator to repress androgen receptor transactivation in prostate cancer cells". Molecular and Cellular Biology. 24 (5): 2202–13. doi:10.1128/MCB.24.5.2202-2213.2004. PMC 350564. PMID 14966297.
- ^ Rao MA, Cheng H, Quayle AN, Nishitani H, Nelson CC, Rennie PS (December 2002). "RanBPM, a nuclear protein that interacts with and regulates transcriptional activity of androgen receptor and glucocorticoid receptor". The Journal of Biological Chemistry. 277 (50): 48020–7. doi:10.1074/jbc.M209741200. PMID 12361945.
- ^ Beitel LK, Elhaji YA, Lumbroso R, Wing SS, Panet-Raymond V, Gottlieb B, et al. (August 2002). "Cloning and characterization of an androgen receptor N-terminal-interacting protein with ubiquitin-protein ligase activity". Journal of Molecular Endocrinology. 29 (1): 41–60. doi:10.1677/jme.0.0290041. PMID 12200228.
- ^ Lu J, Danielsen M (November 1998). "Differential regulation of androgen and glucocorticoid receptors by retinoblastoma protein". The Journal of Biological Chemistry. 273 (47): 31528–33. doi:10.1074/jbc.273.47.31528. PMID 9813067.
- ^ Yeh S, Miyamoto H, Nishimura K, Kang H, Ludlow J, Hsiao P, et al. (July 1998). "Retinoblastoma, a tumor suppressor, is a coactivator for the androgen receptor in human prostate cancer DU145 cells". Biochemical and Biophysical Research Communications. 248 (2): 361–7. doi:10.1006/bbrc.1998.8974. PMID 9675141.
- ^ Miyamoto H, Rahman M, Takatera H, Kang HY, Yeh S, Chang HC, et al. (February 2002). "A dominant-negative mutant of androgen receptor coregulator ARA54 inhibits androgen receptor-mediated prostate cancer growth". The Journal of Biological Chemistry. 277 (7): 4609–17. doi:10.1074/jbc.M108312200. PMID 11673464.
- ^ Kang HY, Yeh S, Fujimoto N, Chang C (March 1999). "Cloning and characterization of human prostate coactivator ARA54, a novel protein that associates with the androgen receptor". The Journal of Biological Chemistry. 274 (13): 8570–6. doi:10.1074/jbc.274.13.8570. PMID 10085091.
- ^ Moilanen AM, Poukka H, Karvonen U, Häkli M, Jänne OA, Palvimo JJ (September 1998). "Identification of a novel RING finger protein as a coregulator in steroid receptor-mediated gene transcription". Molecular and Cellular Biology. 18 (9): 5128–39. doi:10.1128/mcb.18.9.5128. PMC 109098. PMID 9710597.
- ^ Poukka H, Aarnisalo P, Santti H, Jänne OA, Palvimo JJ (January 2000). "Coregulator small nuclear RING finger protein (SNURF) enhances Sp1- and steroid receptor-mediated transcription by different mechanisms". The Journal of Biological Chemistry. 275 (1): 571–9. doi:10.1074/jbc.275.1.571. PMID 10617653.
- ^ Liu Y, Kim BO, Kao C, Jung C, Dalton JT, He JJ (May 2004). "Tip110, the human immunodeficiency virus type 1 (HIV-1) Tat-interacting protein of 110 kDa as a negative regulator of androgen receptor (AR) transcriptional activation". The Journal of Biological Chemistry. 279 (21): 21766–73. doi:10.1074/jbc.M314321200. PMID 15031286.
- ^ Chipuk JE, Cornelius SC, Pultz NJ, Jorgensen JS, Bonham MJ, Kim SJ, et al. (January 2002). "The androgen receptor represses transforming growth factor-beta signaling through interaction with Smad3". The Journal of Biological Chemistry. 277 (2): 1240–8. doi:10.1074/jbc.M108855200. PMID 11707452.
- ^ Hayes SA, Zarnegar M, Sharma M, Yang F, Peehl DM, ten Dijke P, et al. (March 2001). "SMAD3 represses androgen receptor-mediated transcription". Cancer Research. 61 (5): 2112–8. PMID 11280774.
- ^ Kang HY, Huang KE, Chang SY, Ma WL, Lin WJ, Chang C (November 2002). "Differential modulation of androgen receptor-mediated transactivation by Smad3 and tumor suppressor Smad4". The Journal of Biological Chemistry. 277 (46): 43749–56. doi:10.1074/jbc.M205603200. PMID 12226080.
- ^ Gobinet J, Auzou G, Nicolas JC, Sultan C, Jalaguier S (December 2001). "Characterization of the interaction between androgen receptor and a new transcriptional inhibitor, SHP". Biochemistry. 40 (50): 15369–77. doi:10.1021/bi011384o. PMID 11735420.
- ^ Unni E, Sun S, Nan B, McPhaul MJ, Cheskis B, Mancini MA, et al. (October 2004). "Changes in androgen receptor nongenotropic signaling correlate with transition of LNCaP cells to androgen independence". Cancer Research. 64 (19): 7156–68. doi:10.1158/0008-5472.CAN-04-1121. PMID 15466214.
- ^ Powell SM, Christiaens V, Voulgaraki D, Waxman J, Claessens F, Bevan CL (March 2004). "Mechanisms of androgen receptor signalling via steroid receptor coactivator-1 in prostate". Endocrine-Related Cancer. 11 (1): 117–30. doi:10.1677/erc.0.0110117. PMID 15027889.
- ^ Yuan X, Lu ML, Li T, Balk SP (December 2001). "SRY interacts with and negatively regulates androgen receptor transcriptional activity". The Journal of Biological Chemistry. 276 (49): 46647–54. doi:10.1074/jbc.M108404200. PMID 11585838.
- ^ Matsuda T, Junicho A, Yamamoto T, Kishi H, Korkmaz K, Saatcioglu F, et al. (April 2001). "Cross-talk between signal transducer and activator of transcription 3 and androgen receptor signaling in prostate carcinoma cells". Biochemical and Biophysical Research Communications. 283 (1): 179–87. doi:10.1006/bbrc.2001.4758. PMID 11322786.
- ^ Ueda T, Bruchovsky N, Sadar MD (March 2002). "Activation of the androgen receptor N-terminal domain by interleukin-6 via MAPK and STAT3 signal transduction pathways". The Journal of Biological Chemistry. 277 (9): 7076–85. doi:10.1074/jbc.M108255200. PMID 11751884.
- ^ Ting HJ, Yeh S, Nishimura K, Chang C (January 2002). "Supervillin associates with androgen receptor and modulates its transcriptional activity". Proceedings of the National Academy of Sciences of the United States of America. 99 (2): 661–6. Bibcode:2002PNAS...99..661T. doi:10.1073/pnas.022469899. PMC 117362. PMID 11792840.
- ^ Mu X, Chang C (October 2003). "TR2 orphan receptor functions as negative modulator for androgen receptor in prostate cancer cells PC-3". The Prostate. 57 (2): 129–33. doi:10.1002/pros.10282. PMID 12949936. S2CID 24134119.
- ^ Lee YF, Shyr CR, Thin TH, Lin WJ, Chang C (December 1999). "Convergence of two repressors through heterodimer formation of androgen receptor and testicular orphan receptor-4: a unique signaling pathway in the steroid receptor superfamily". Proceedings of the National Academy of Sciences of the United States of America. 96 (26): 14724–9. Bibcode:1999PNAS...9614724L. doi:10.1073/pnas.96.26.14724. PMC 24715. PMID 10611280.
- ^ Wang X, Yang Y, Guo X, Sampson ER, Hsu CL, Tsai MY, et al. (May 2002). "Suppression of androgen receptor transactivation by Pyk2 via interaction and phosphorylation of the ARA55 coregulator". The Journal of Biological Chemistry. 277 (18): 15426–31. doi:10.1074/jbc.M111218200. PMID 11856738.
- ^ Hsiao PW, Chang C (August 1999). "Isolation and characterization of ARA160 as the first androgen receptor N-terminal-associated coactivator in human prostate cells". The Journal of Biological Chemistry. 274 (32): 22373–9. doi:10.1074/jbc.274.32.22373. PMID 10428808.
- ^ Miyajima N, Maruyama S, Bohgaki M, Kano S, Shigemura M, Shinohara N, et al. (May 2008). "TRIM68 regulates ligand-dependent transcription of androgen receptor in prostate cancer cells". Cancer Research. 68 (9): 3486–94. doi:10.1158/0008-5472.CAN-07-6059. PMID 18451177.
- ^ Poukka H, Aarnisalo P, Karvonen U, Palvimo JJ, Jänne OA (July 1999). "Ubc9 interacts with the androgen receptor and activates receptor-dependent transcription". The Journal of Biological Chemistry. 274 (27): 19441–6. doi:10.1074/jbc.274.27.19441. PMID 10383460.
- ^ Müller JM, Isele U, Metzger E, Rempel A, Moser M, Pscherer A, et al. (February 2000). "FHL2, a novel tissue-specific coactivator of the androgen receptor". The EMBO Journal. 19 (3): 359–69. doi:10.1093/emboj/19.3.359. PMC 305573. PMID 10654935.
- ^ Cheng S, Brzostek S, Lee SR, Hollenberg AN, Balk SP (July 2002). "Inhibition of the dihydrotestosterone-activated androgen receptor by nuclear receptor corepressor". Molecular Endocrinology. 16 (7): 1492–501. doi:10.1210/mend.16.7.0870. PMID 12089345.
- ^ Hodgson MC, Astapova I, Cheng S, Lee LJ, Verhoeven MC, Choi E, et al. (February 2005). "The androgen receptor recruits nuclear receptor CoRepressor (N-CoR) in the presence of mifepristone via its N and C termini revealing a novel molecular mechanism for androgen receptor antagonists". The Journal of Biological Chemistry. 280 (8): 6511–9. doi:10.1074/jbc.M408972200. PMID 15598662.
- ^ Markus SM, Taneja SS, Logan SK, Li W, Ha S, Hittelman AB, et al. (February 2002). "Identification and characterization of ART-27, a novel coactivator for the androgen receptor N terminus". Molecular Biology of the Cell. 13 (2): 670–82. doi:10.1091/mbc.01-10-0513. PMC 65658. PMID 11854421.
- ^ Sharma M, Li X, Wang Y, Zarnegar M, Huang CY, Palvimo JJ, et al. (November 2003). "hZimp10 is an androgen receptor co-activator and forms a complex with SUMO-1 at replication foci". The EMBO Journal. 22 (22): 6101–14. doi:10.1093/emboj/cdg585. PMC 275443. PMID 14609956.
Further reading
[edit]- Brinkmann AO (November 2009). "Androgen physiology: receptor and metabolic disorders" (PDF). In McLachlan R (ed.). Endocrinology of Male Reproduction. Endotext.org. Archived from the original (PDF) on 2012-02-22. Retrieved 2008-04-29.
- Gottlieb B (2007-07-24). "The Androgen Receptor Gene Mutations Database Server". McGill University. Archived from the original on 22 April 2008. Retrieved 2008-04-29.
- Thompson J (2006-09-30). Molecular Mechanisms of Androgen Receptor Interactions (PDF) (Ph.D. thesis). University of Helsinki. Biomedical Dissertations No. 80. Archived from the original (PDF) on 6 April 2008. Retrieved 2008-04-29.
External links
[edit]- GeneReviews/NCBI/NIH/UW entry on Androgen Insensitivity Syndrome
- OMIM entries on Androgen Insensitivity Syndrome
- GeneReviews/NIH/NCBI/UW entry on Spinal and Bulbar Muscular Atrophy, Kennedy's Disease, SBMA, X-Linked Spinal and Bulbar Muscular Atrophy
- OMIM entries on Spinal and Bulbar Muscular Atrophy, Kennedy's Disease, SBMA, X-Linked Spinal and Bulbar Muscular Atrophy
- Androgen+Receptors at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Human AR genome location and AR gene details page in the UCSC Genome Browser.
- Overview of all the structural information available in the PDB for UniProt: P10275 (Human Androgen receptor) at the PDBe-KB.
- Overview of all the structural information available in the PDB for UniProt: P19091 (Mouse Androgen receptor) at the PDBe-KB.